Radioligand Therapy Market Size & Share Analysis - Trends, Drivers, Competitive Landscape, and Forecasts (2024 - 2030)
Get a Comprehensive Overview of the Radioligand Therapy Market Report Prepared by P&S Intelligence, Segmented by Indication (Prostate Cancer, Neuroendocrine Tumors), Drug (Lutathera, Pluvicto, Xtandi, Xofigo, Zytiga), End User (Hospitals, Clinics, Academic and Research Institutes), Distribution Channel (Direct, Indirect), and Geographic Regions. This Report Provides Insights From 2019 to 2030.
Radioligand Therapy Market Size
Market Statistics
| Study Period | 2019 - 2030 |
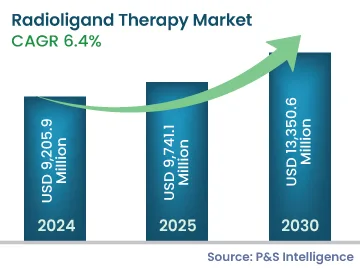
| 2024 Market Size | USD 9,205.9 Million |
| 2025 Market Size | USD 9,741.1 Million |
| 2030 Forecast | USD 13,350.6 Million |
| Growth Rate (CAGR) | 6.4% |
| Largest Region | North America |
| Fastest-Growing Region | Asia-Pacific |
| Nature of the Market | Fragmented |
| Largest End User Category | Hospitals |
Market Size Comparison
Key Players

Key Report Highlights
|
Explore the market potential with our data-driven report
Radioligand Therapy Market Analysis
The radioligand therapy market size is estimated to have stood at USD 9,205.9 million in 2024, and rising at a compound annual growth rate of 6.4% during 2024–2030, it will reach USD 9,741.1 million by 2030. The market for radioligand therapy is driven by the increasing number of FDA-approved drugs with low side-effects. The progress is augmented by the increasing research on cancer treatment and the rising demand for more-effective drugs. Nuclear medicine physicians are encouraging the establishment of centralized radio-pharmacies, as radiopharmaceuticals are the major area of interest for researchers, for targeted therapy. For instance, in the U.S., Cardinal Health and Jubilant Radio Pharma are the key players in the radio-pharmacies market.
Moreover, governments are taking initiatives and raising funds to reduce the cancer burden. For instance, the European Commission launched Europe’s Beating Cancer Plan in February 2021. The plan aims to provide cancer services with an emphasis on disease prevention and early diagnosis. In 2022, the European government has planned to pour more than USD 4.2 billion into cancer research and care. Radiopharmaceuticals and radiotherapy are highly effective and are being, thus, demanded increasingly for malignancies, which augments growth.
For instance, the POINT Biopharma Global Inc. is preparing to enter the market for radioligand therapy with PNT2003 drug, which is currently in the final phases of clinical trials. Similarly, considering the increasing demand for radiotherapy and government funding in this field, market players are strengthening their focus on the development, approval, and launch of advanced products, especially drugs based on radioligands.
Radioligand Therapy Market Trends & Growth Drivers
Developments in Technology Are Biggest Market Trends
- In response to the increasing demand for cancer therapies, major corporations are increasing their investments in the development of improved diagnostics, drugs, equipment, technologies, and treatment alternatives.
- The increasing popularity of radioligand treatment offers research provides more avenues for market players to capitalize on its potential.
- The emergence of contract research organizations has presented novel options for market participants, since it has allowed them to provide enhanced services at more affordable prices and with better expertise.
- The amount of money provided by the government to upgrade public healthcare facilities has increased, encouraging greater research into more efficient treatment options.
- Prominent industry players are joining forces to create innovative technologies for targeted disease diagnosis, in order to capture a sizeable portion of the market in developing countries.
Rising Prevalence of Prostate Cancer Propels Radioligand Therapy Demand
- The radioligand therapy market trends for prostate cancer and neuroendocrine tumors (NET) are the most promising.
- In the U.S., prostate cancer is the second-leading cause of cancer death in men.
- Similarly, more than 12,000 people in the U.S. are diagnosed with a NET each year, with the count increasing every twelvemonth. The increase in prostate cancer and NET cases will drive the demand for diagnoses, including better imaging tests and endoscopy procedures. In turn, accurate diagnoses will drive the adoption of radioligand therapy.
- The increase in prostate cancer and NET cases will drive the demand for diagnosis, including better imaging tests and endoscopy procedures; thus, the rise in the awareness of these tumors drive the adoption of radioligand therapy.
Rising Rates of Cancer Are Major Driver for Market
- The biggest driver for the radioligand therapy market is the growing prevalence of cancer, primarily due to the increase in life expectancy.
- Nonetheless, people are more likely to get cancer as they get older. As a result, there are more cancer patients worldwide due to the aging population. Furthermore, since obesity is associated with a higher risk of malignancies, contemporary lifestyles, characterized by extended work hours and sedentary behaviors, in addition to bad dietary patterns which include highly processed, sugary foods and beverages, contribute to a higher risk of cancer.
- The number of people with diabetes is growing, which raises the incidence of cancer cases. However, with the extensive R&D being carried out in oncology, cancer therapies have diversified and advanced.
- Among these developments, radioligand therapy has become a popular choice as it delivers radiation energy directly to the tumor cells, in an effort to destroy them in situ.
Lack of Suitable Infrastructure in Developing Countries Hampers Market Expansion
- In low- and middle-income countries, the lack of suitable infrastructure poses a significant obstacle to market expansion. The high costs associated with R&D and therapy equipment, limited infrastructure, and low awareness levels are expected to impede market growth.
- The absence of favorable reimbursement scenarios and limited technology penetration in developing economies, and budget cuts in healthcare facilities—especially during the pandemic would further exacerbate the challenges faced by the market during the forecast period.
Strict Safety Guidelines and Complex Approval Process Hamper Market Growth
- To protect consumer security and best interests, several medications, therapies, and treatment plans are rigorously evaluated, tested, and subjected to several levels of safety regulations and monitoring.
- Before being certified for use in clinical situations in hospitals and clinics, radioligand treatment and its subsequent developments must pass a number of stringent tests.
- In the case of a successful trials, the remedies still need to be approved by the relevant authorities. For the same, the authorities uphold strict accountability and compliance norms. The examination process takes time for the market players.
- Market participants lose a great deal of time as they wait for permission, which is periodically delayed by administrative problems.
Expanding Focus on Radiopharmaceuticals Is an Opportunity
- The improvements in nuclear medicine are expected to be the key enabler for radiopharmaceuticals to obtain a major place in medicine. Radiopharmaceuticals are being increasingly used in the diagnosis and treatment of several diseases, such as cancer and neurological disorders.
- Moreover, several radio drugs have been approved by the FDA, such as Zytiga (launched in 2017), Lutathera (launched in 2018), and Pluvicto (launched in 2022).
- The frequent approvals will improve the potential for radiotherapy and increase the opportunities for established as well as emerging players, especially considering that the currently available medicines have already been accepted by more than 100 countries.
- Owing to their radioactive decay and targeted therapeutic irradiation, radioligands, or radiopharmaceuticals, are extensively used for cancer treatment.
Radioligand Therapy Industry Outlook
Indication Insights
- With almost 55% of the market share in 2024, the prostate cancer category dominating the market. Males are the victims of prostate cancer, which affects the prostate glands in the male reproductive system.
- About 1 million new cases of prostate cancer are identified worldwide each year, making it a serious global health concern. Prostate cancer affects roughly 13 out of every 100 men in the U.S, and accounts for 1 in every 40 male fatalities. Thus, it is given considerable attention in the field of oncology due to its substantial impact.
- Because of the high precision and success rate of radioligand therapy, it has become a popular therapeutic choice for prostate cancer. Patients now have significantly higher survival rates because of this therapy.
The indications covered in the report include:
- Prostate Cancer (Largest and Fastest-Growing Category)
- Neuroendocrine Tumors (NETs)
- Others
Drug Insights
- Lutathera is the largest category, with an estimated share of 30% in 2024. It is because it is the first approved radiopharmaceutical for PRRT.
- Pluvicto is the fastest-growing category as it received the FDA’s approval in March 2022 for treating progressive, PSMA-positive metastatic castration-resistant prostate cancer.
The following drugs are included in the report:
- Lutathera (Largest Category)
- Pluvicto (Fastest-Growing Category)
- Xtandi
- Xofigo
- Zytiga
- Others
End User Insights
- Hospitals holds the largest share in the end user segment, of 45%, in 2024. Hospitals are the first choice of patients for long-term cancer therapy, and they have the finances to afford the expensive RLT equipment.
- Investments in the healthcare infrastructure are projected to increase, which will further support hospitals' dominance on the global market for radioligand therapy. oncology departments frequently focus on giving patients comprehensive therapies that are specific to their requirements. Utilizing their specialized infrastructure and knowledge, hospitals serve as vital hubs for the delivery of the radioligand therapy.
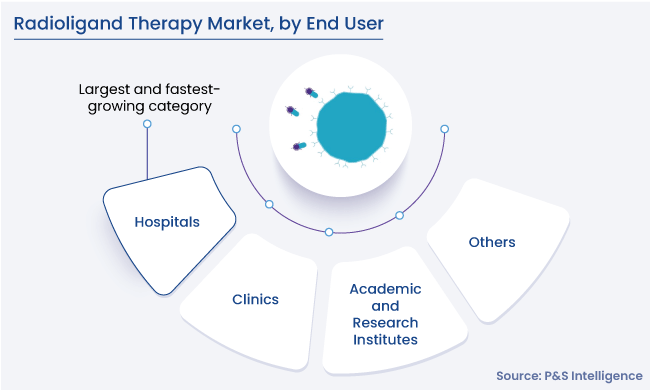
The end users covered in the report include:
- Hospitals (Largest and Fastest-Growing Category)
- Clinics
- Academic and Research Institutes
- Others
Distribution Channel Insights
The indirect category holds the largest share, of 70%, in 2024, and it is also expected to witness the highest CAGR, of 6.6%, over the forecast period. Pharmaceutical manufacturers sell to drug wholesalers, from whom pharmacies purchase the medicines and finally sell them to the patient.
The distribution channels covered in the report include:
- Direct
- Indirect (Larger and Faster-Growing Category)
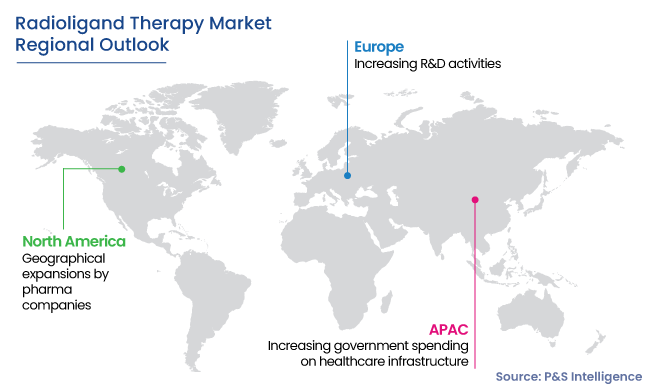
North America Is Largest Market
- North America is dominating the market in 2024 with a share of 50%. The domain advance here is majorly augmented by the FDA’s approval to an increasing number of drugs, geographical expansions by pharma companies, and rising government funding for cancer research.
- The market growth in the U.S. is attributed to the government initiatives for improving chronic disease care and rising number of specialized cancer hospitals and research centers. Moreover, programs such as the Cancer Moonshot Program aim to reduce the count of deaths due to cancer.
- The Asia-Pacific region is expected to witness the fastest growth during the forecast period, of 6.7%. This can be attributed to increasing government spending on healthcare infrastructure and heightening R&D activities undertaken by the leading market players in the region.
The regions and countries analyzed in this report include:
- North America (Largest Regional Market)
- U.S. (Larger and Faster-Growing Country Market)
- Canada
- Europe
- Germany (Largest Country Market)
- U.K. (Fastest-Growing Country Market)
- France
- Italy
- Spain
- Rest of Europe
- Asia-Pacific (APAC) (Fastest-Growing Regional Market)
- China (Largest Country Market)
- Japan
- India (Fastest-Growing Country Market)
- South Korea
- Australia
- Rest of APAC
- Latin America (LATAM)
- Brazil (Largest and Fastest-Growing Country Market)
- Mexico
- Rest of LATAM
- Middle East and Africa (MEA)
- Saudi Arabia (Largest Country Market)
- South Africa
- U.A.E. (Fastest-Growing Country Market)
- Rest of MEA
Market Nature – Fragmented
- The radioligand therapy market is fragmented with the presence of several key players. Pharma firms have been involved in product launches and partnerships to remain competitive in the market.
- Moreover, the players in the market for radiopharmaceuticals are collaborating to better cater to the rising demand for radioligand drugs. Thus, a few targeted cancer therapies using radioactive or nuclear agents are in the final phases of clinical trials, while a few more are in the research phase.
- The current market for radioligand therapy is majorly dominated by manufacturers such as Novartis International AG, POINT Biopharma Global Inc., Telix Pharmaceuticals, Bayer AG, Molecular Partners, ITM Isotope Technologies Munich SE, Radio Medix, Pfizer Inc., and Janssen Global Services LLC.
Top Radioligand Therapy Companies:
- Novartis International AG
- POINT Biopharma Global Inc.
- Telix Pharmaceuticals
- Bayer AG
- Convergent Therapeutics
- Eli Lilly and Company
- Molecular Partners
- ITM Isotope Technologies Munich SE
- Pfizer Inc.
- Janssen Global Services LLC
- Fusion Pharmaceuticals
Radioligand Therapy Industry News
- In June 2022, Fusion Pharmaceuticals Inc., a clinical-stage oncology company, provided updates on its Phase 1 clinical trials of the radiopharmaceuticals FPI-1434 and FPI-1966.
- In March 2022, Pluvicto by Novartis was approved by the FDA as a targeted radioligand therapy for progressive PSMA-positive metastatic castration-resistant prostate cancer.
- In October 2023, Eli Lilly and Company announced plans for the acquisition of POINT Biopharma to develop next-generation radioligand therapies for cancer.
- In December 2023, Pfizer Inc. completed the acquisition of Seagen, a global biotechnology company that discovers, develops, and commercializes next-generation cancer medicines.
- In May 2023, Australia-based Telix Pharmaceuticals Limited announced an agreement with Bayer AG to supply Illuccix (TLX591-CDx) kit for the preparation of gallium Ga 68 gozetotide injection.
- In December 2021, Molecular Partners announced a collaboration with Novartis to develop Darpin-conjugated radioligand therapeutic candidates for oncology.
Request the Free Sample Pages

