Wound Care Biologics Market to Generate Revenue Worth $2 Billion by 2024
- Published: December 2019
The global wound care biologics market was valued at $1.4 billion in 2018 and is projected to reach $2.0 billion by 2024, progressing at a CAGR of 7.0% during the forecast period (2019–2024). Among the product type, the skin substitutes category held the largest revenue share in the market in 2018. It is also expected to witness the fastest growth during the forecast period. The market growth is mainly driven by rise in incidence of burn injuries, surge in geriatric population, introduction of innovative wound care biologics products, awareness programs for wound care treatment and management, and increase in research and development activities.
Potential Applications of Stem Cell Therapy is a Key Trend Observed in the Wound Care Biologics Market
Research studies evaluating novel stem cell-based therapeutics to limit the requirement of repetitive treatment is a key trend observed in the wound care biologics market. For instance, RenovaCare Inc.’s CellMist solution is a liquid suspension that contains patient’s own regenerative skin stem cells. The liquid solution is placed in a SkinGun that sprays the cells onto the wound sites to promote healing of burns and other wounds. Therefore, the use of stem cells has demonstrated considerable potential for improving the rate and quality of wound healing and regenerating the skin.
Wound Care Biologics Market is Likely to Witness Significant Growth in the Coming Years Mainly Due to Surging Geriatric Population
Globally, surge in geriatric population has led to high healthcare expenditure, which is driving the growth of the wound care biologics market. This is mainly due to the fact that elderly patients require extensive care as they are prone to illness, due to low immunity levels. In addition, aged people have weakened body functions, such as impaired blood circulation and reduced collagen deposition, which require longer recovery period for wounds to heal, in comparison to adults. According to the United Nations Department of Economic and Social Affairs (UNDESA) report on the World Population Aging, the population of people aged 60 years and above is growing at a high rate. The number of people aged 60 years and above increased from 382 million in 1980 to 962 million in 2017, and is expected to reach 2.1 billion by 2050.
Segmentation Analysis of the Wound Care Biologics Market
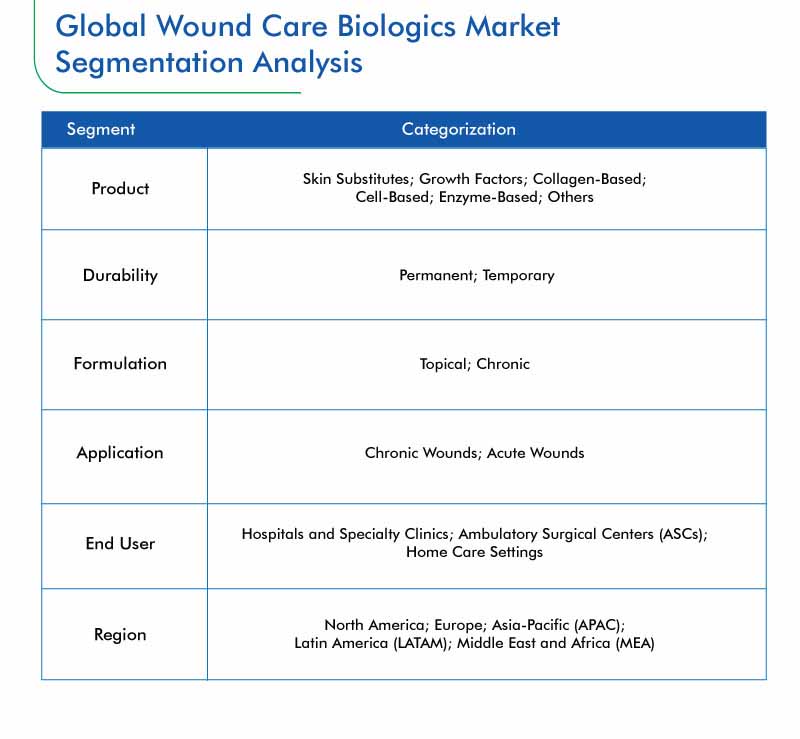
- Based on product, the skin substitutes category is expected to witness the fastest growth during the forecast period, globally. This is mainly due to the fact that these products have an intact extracellular matrix structure and allow the construction of natural dermis. In addition, skin substitutes possess characteristics of re-epithelialisation.
- On the basis of durability, the permanent category holds larger revenue share in the wound care biologics market. This is mainly attributed to the increasing prevalence of chronic wounds, and improved durable nature of permanent biologics compared to temporary biologics.
- Topical is faster growing formulation category, mainly due to the fact that topical wound care biologics have rapid and improved wound closure properties compared to other formulations.
- Based on application, chronic wounds generate larger revenue in the wound care biologics market. This is mainly due to the fact that the prevalence of chronic wounds, including diabetic foot ulcers (DFUs), pressure ulcers, and venous leg ulcers (VLUs), is rising at a faster rate compared to acute wounds across the globe. Moreover, cost for treating chronic wounds is high, thereby increasing its overall revenue contribution to the market.
- Wound care biologics are largely being used in hospitals and specialty centers. This end-user category held the largest revenue share, of 81.3%, in 2018 in the wound care biologics market. This is ascribed to the fact that patients with chronic wounds require extensive care under the supervision of trained healthcare professionals.
Geographical Analysis of the Wound Care Biologics Market
Together, North America and APAC are expected to account for over 65% revenue share in the global wound care biologics market by 2024. This can be mainly attributed to the presence of large number of market players, high healthcare spending, and increasing research and development (R&D) activities taking place in these regions.
Browse report overview with detailed TOC on "Wound Care Biologics Market Research Report: By Product (Skin Substitutes, Growth Factors, Collagen-Based, Cell-Based, Enzyme-Based), Durability (Permanent, Temporary), Formulation (Topical), Application (Chronic, Acute), End-User (Hospitals & Specialty Clinics, ASCs, Home Care), Geographical Outlook (U.S., Canada, Germany, France, U.K., Italy, Spain, China, Japan, India, Australia, Mexico, Brazil, Saudi Arabia, South Africa, Israel) – Industry Size and Share Analysis and Growth Forecast to 2024" at:https://www.psmarketresearch.com/market-analysis/wound-care-biologics-market
Other geographies, such as Europe, LATAM, and MEA, also hold considerable revenue shares in the wound care biologics market, on account of rising incidence of burns and traumatic injuries, surging geriatric population, and large population base in these regions.
- Germany is expected to lead the European wound care biologics market, generating revenue of over $124.1 million by 2024. This can be mainly attributed to the technological advancements and increasing awareness about wound care biologics for the treatment of acute and chronic wounds in the country.
- Mexico is the fastest-growing market for wound care biologics in the LATAM region. It is mainly driven by increasing awareness among people to heal chronic wounds and burn injuries using human- and animal-based biologics as they have high collagen content.
- Due to the innovation of wound care biologic products and rapid urbanization, Saudi Arabia is expected to dominate the MEA wound care biologics market by 2024, with a revenue share of 34.0%.
Competitive Landscape of the Wound Care Biologics Market
Mölnlycke Health Care AB, Integra LifeSciences Corporation, MiMedx Group Inc., Osiris Therapeutics Inc., Smith & Nephew plc, Organogenesis, and Acell Inc. are some of the major players operating in the wound care biologics industry.
In May 2019, MediWound Ltd., an integrated biotechnology company, entered into an exclusive licensing and supply agreements with Vericel Corporation to commercialize NexoBrid, a topically-administered biologic product that removes eschar in patients with deep partial and full-thickness thermal burns, in North America. Under the terms of the agreement, MediWound Ltd. will be responsible for the development and commercialization activities of NexoBrid and to obtain the U.S. marketing approval from the United States Food and Drug Administration (USFDA). Some other important players operating in the wound care biologics market are Wright Medical Group N.V., Anika Therapeutics Inc., Vericel Corporation, MediWound Ltd., and RenovaCare Inc.
Market Segmentation by Product
-
Skin Substitutes
- Human donor-tissue derived
- Acellular animal-derived
- Biosynthetic
-
Growth Factors
- Platelet-derived growth factors (PDGF)
- Fibroblast growth factors (FGF)
- Epidermal growth factors (EGF)
- Transforming growth factor-beta (TGF-β)
- Insulin-like growth factors (IGF)
- Others
- Collagen-Based
-
Cell-Based
- Viable
- Non-viable
- Enzyme-Based
- Others
Market Segmentation by Durability
- Permanent
- Temporary
Market Segmentation by Formulation
- Topical
- Others
Market Segmentation by Application
-
Chronic
- Diabetic foot ulcers (DFUs)
- Pressure ulcers
- Venous leg ulcers (VLU)
-
Acute
- Surgical, traumatic, and burn injuries
- Others
Market Segmentation by End User
-
Hospitals & Specialty Clinics
- Inpatient settings
- Outpatient settings
- ASCs
- Home Care
Market Segmentation by Geography
-
North America Wound Care Biologics Market
- By product
- By durability
- By formulation
- By application
- By end user
-
By country – U.S. and Canada
-
U.S. wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Canada wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
U.S. wound care biologics market
-
Europe Wound Care Biologics Market
- By product
- By durability
- By formulation
- By application
- By end user
-
By country – Germany, France, U.K., Italy, Spain, and Rest of Europe
-
Germany wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
France wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
U.K. wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Italy wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Spain wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Germany wound care biologics market
-
Asia-Pacific (APAC) Wound Care Biologics Market
- By product
- By durability
- By formulation
- By application
- By end user
-
By country – China, Japan, India, Australia, and Rest of APAC
-
China wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Japan wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
India wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Australia wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
China wound care biologics market
-
Latin America (LATAM) Wound Care Biologics Market
- By product
- By durability
- By formulation
- By application
- By end user
-
By country – Mexico, Brazil, and Rest of LATAM
-
Mexico wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Brazil wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Mexico wound care biologics market
-
Middle East & Africa (MEA) Wound Care Biologics Market
- By product
- By durability
- By formulation
- By application
- By end user
-
By country – Saudi Arabia, South Africa, Israel, and Rest of MEA
-
Saudi Arabia wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
South Africa wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Israel wound care biologics market
- By product
- By durability
- By formulation
- By application
- By end user
-
Saudi Arabia wound care biologics market