Report Code: 10603 | Available Format: PDF | Pages: 77
U.S. Scar Treatment Market by Type of Treatment (Topical [Cream, Oil, Gel, Gel Sheet], Surface [Dermabrasion, Chemical Peeling, Cryosurgery], Laser [CO2, Erbium: YAG, Pulsed-Dye], Injectable [Tissue/Dermal Filler Injection, Steroid Injection], Surgical), by Type of Scar (Post-Surgical, Acne, Keloid & Hypertrophic, Contracture, Stretch Mark), by End User (Home-Use, Hospitals, Dermatology Clinics) - Market Size, Share, Development, Growth, and Demand Forecast, 2013-2023
- Report Code: 10603
- Available Format: PDF
- Pages: 77
- Report Description
- Table of Contents
- Market Segmentation
- Request Free Sample
U.S. Scar Treatment Market Overview
The U.S. scar treatment market generated $6.3 billion revenue in 2017 and is projected to witness a CAGR of 10.4% during the 2018 - 2023, due to increasing number of surgeries for the improvement of physical appearance, growing prevalence of skin problems that lead to scar formation, increasing expenditure on personal care, and technological advancements in dermatological products.
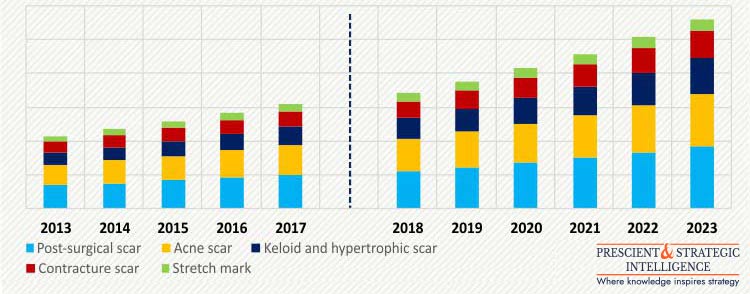
U.S. SCAR TREATMENT MARKET, BY TYPE OF SCAR, $M (2013-2023)
The scar treatment market in the U.S. has been categorized as topical, surface, laser, injectable, and surgical, on the basis of type of treatment. Among these, the topical scar treatment category accounted for the largest share in this market, with 61.2% contribution in 2017. This is mainly due to the increasing demand for topical products such as creams, oils, gels, and gel sheets in the country. Recently, silicon gels are being widely used for treatment of scars. These gels prevent scar formation in freshly healed wounds and also reduce prominent immature scars.
The scar treatment market in the U.S. is categorized into post-surgical scar, acne scar, keloid and hypertrophic scar, contracture scar, and stretch mark, on the basis of type of scar. The demand for products to treat keloid and hypertrophic scars is expected to witness fastest growth during the forecast period, at a CAGR of 11.6%. This growth is attributable to the increasing number of burn cases, general surgical procedures, and chronic wounds since these are the major reasons for the formation of such scars.
According to the U.S. National Institutes of Health, in the U.S. nearly 6.5 million people suffered from chronic wound in 2017, which in turn is anticipated to result in increased cases of hypertrophic scars.
Among different end users, during the historical period, home-use held the largest share as an end-user category in the scar treatment market of the U.S., with 36.8% share in 2017. This is attributable to the increasing disposable income of general population leading to high expenditure on personal care, and growing adoption of home-use topical products such as gels, creams and oils for removal or reduction of scars, due to their low cost and increasing efficacy.
U.S. Scar Treatment Market Dynamics
Growth Drivers
Increasing prevalence of skin diseases is a key driver leading to the growth of the scar treatment market in the U.S. Acne is the most common dermatological condition, which is caused due to clogging of hair follicles by dirt, sebum, and dead skin cells.
Furthermore, acne affects majority of the world’s population and can lead to scaring. According to the American Academy of Dermatology (AAD), approximately 85% of the U.S. population suffers from acne at some phase of their lives, which eventually lead to scar formation. Burns are another major reason for scar formation. According to the American Burn Association (ABA), in 2011, approximately 486,000 burn injuries were reported in the U.S.
Moreover, the expenditure on personal care is increasing in the U.S., which in turn, is contributing to the growth of the U.S. market for scar treatment. According to the AAD, 40 to 50 million Americans spend $2 million every year for the treatment and prevention of acne. Due to this increasing awareness about various scar treatments, the expenditure on treating them has also been increasing.
U.S. Scar Treatment Market Competitive Landscape
The companies operating in the U.S. scar treatment industry are launching topical products such as gels and creams to expand their market share in the home-use category. For instance, in June 2017, Sonoma Pharmaceutical Inc announced the Singapore approval for Microcyn (hypochlorous acid) for the treatment of atopic dermatitis and hypertrophic /keloid scars. One of these approvals was received for EZyma hydrogel, which is used for the treatment of itching dryness, redness, burning and pain associated with atopic dermatitis.
Similarly, in March 2017, Sonoma Pharmaceuticals Inc, received U.S. Food and Drug Administration (FDA) approval for its new product, Loyon skin descale, for the treatment of skin scaling related to dermatoses. It can be easily applied and spread into the cracks owing to lower surface tension.
Some of the other key players operating in the U.S. scar treatment market are Revitol Corporation Inc., CCA Industries Inc., Scarguard Labs LLC, Quantum Health, Valeant Pharmaceuticals International Inc., Pacific World Corporation, Merz Pharma GmbH & Co. KGaA, and Johnson & Johnson.
Want a report tailored exactly to your business strategy?
Request CustomizationWant an insight-rich discussion with the report author?
Speak to AnalystOur dedication to providing the most-accurate market information has earned us verification by Dun & Bradstreet (D&B). We strive for quality checking of the highest level to enable data-driven decision making for you
Our insights into the minutest levels of the markets, including the latest trends and competitive landscape, give you all the answers you need to take your business to new heights
With 24/7 research support, we ensure that the wheels of your business never stop turning. Don’t let time stand in your way. Get all your queries answered with a simple phone call or email, as and when required
We take a cautious approach to protecting your personal and confidential information. Trust is the strongest bond that connects us and our clients, and trust we build by complying with all international and domestic data protection and privacy laws