Report Code: 11489 | Available Format: PDF | Pages: 177
Surgical Site Infection (SSI) Market by Product (Antibiotic Prophylaxis Products, Antiseptics & Disinfectants, Wound Care Dressings, Surgical Drapes, Surgical Staff Clothing, Patient-Warming Products, Wound Care Supplementary Products, Clippers, Wound Irrigation Products), by Phase (Intraoperative Phase, Preoperative Phase, Postoperative Phase), by End User (Hospitals, Ambulatory Surgical Centers [ASCs], Clinics), by Geography (U.S., Canada, Germany, U.K., France, Italy, Spain, Japan, China, India, Australia, South Korea, Brazil, Mexico, Saudi Arabia, South Africa) – Global Market Size, Share, Development, Growth, and Demand Forecast, 2013–2023
- Report Code: 11489
- Available Format: PDF
- Pages: 177
- Report Description
- Table of Contents
- Market Segmentation
- Request Free Sample
Surgical Site Infection Market Overview
The global surgical site infection market generated $4.0 billion revenue in 2017 and is projected to witness a CAGR of 6.4% during the forecast period, on account of the rising incidence of SSIs, increasing number of surgical procedures, development of technologically advanced products, and growing geriatric population.
GLOBAL SURGICAL SITE INFECTION MARKET, BY PRODUCT, $M (2013–2023)
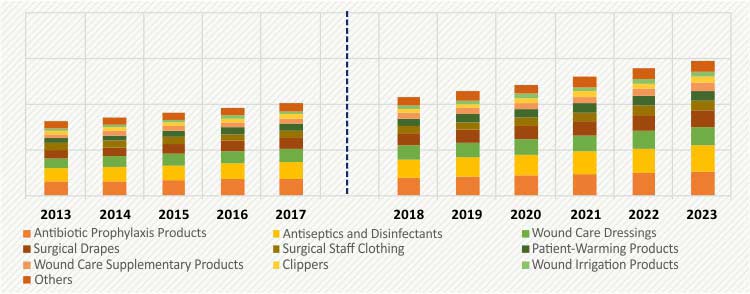
On the basis of product, the surgical site infection market is categorized into antibiotic prophylaxis products, antiseptics and disinfectants, wound care dressings, surgical drapes, surgical staff clothing, patient-warming products, wound care supplementary products, clippers, wound irrigation products, and others. In 2017, antibiotic prophylaxis products generated revenue of $761.1 million, accounting for 18.8% share in the market. Antibiotic prophylaxis products are the most common form of preventive measure that healthcare facilities adopt to avoid infection and contamination during the course of treatment.
The surgical site infection market is also classified into intraoperative, preoperative, and postoperative phases. In terms of value, the intraoperative phase was the largest category in the market, with 40.8% contribution, in 2017. This can be attributed to factors such as the high volume of products consumed to mitigate the chances of infection during this phase and the adherence of healthcare professionals to infection-control guidelines laid down by the World Health Organization (WHO).
On the basis of end user, the surgical site infection market is categorized into hospitals, ambulatory surgery centers (ASCs), clinics, and others. During the forecast period, ASCs are expected to be the fastest-growing category, advancing at a CAGR of 7.1%. This is attributable to the growing preference of ASCs due to increased privacy provided to patients during their stay in these centers.
Globally, North America generated the largest revenue in the surgical site infection market in 2017, with a contribution of 38.4%. The market in the region is expected to witness a CAGR of 6.1% during the forecast period. This can be attributed to the well-developed healthcare industry, presence of leading SSI product manufacturers, and growing awareness on these infections in the region.
Moreover, surge in geriatric population and subsequent increase in the number of surgeries are also propelling the growth of the North American surgical site infection market. According to the UN publication World Population Ageing 2017, around 22.0% of the North American population was above 60 years of age in 2017, and it is expected to increase to 28.0% by 2050.
Surgical Site Infection Market Dynamics
Drivers
Factors such as rising incidence of surgical site infections, increasing number of surgical procedures, development of technologically advanced products, and growing geriatric population are driving the growth of the surgical site infection market. In addition, favorable government regulations are contributing to the growth of the market, globally.
Rising incidence of SSIs is one of the major drivers for the surgical site infection industry. According to the data reported by the European Centre for Disease Prevention and Control (ECDC) on surgical site infection surveillance for 2013–2014, 18,364 infection cases were reported in a total of 967,191 surgical procedures. Furthermore, according to the National Health Surveillance Agency’s report on diagnostic criteria for infections related to health care, SSI is the most prevalent healthcare-related infection in Brazil, ranking third among all infections in health services and constituting 14%–16% of infections found in hospitalized patients.
With the increasing incidence of SSIs, the healthcare expenditure of people is also on the rise. These factors are fueling the demand for the products that help treat these infections, which, in turn, is driving the growth of the surgical site infection market.
Opportunities
Increasing initiatives by various organizations and growing collaborations among major companies to prevent SSIs are offering ample growth oppurtunities to the surgical site infection market players. Various initiatives are being taken by government and non-profit organizations to create awareness on these infections. According to statistics by the WHO, more than 1 in 10 people who undergo surgeries in low- and middle-income countries (LMICs) contract surgical site infections. Further, the risk of acquiring an infection is three to five times higher in patients based in LMICs, as compared to those in high-income countries.
The WHO’s awareness program Patient Safety Solutions mandates adherence to hand hygiene in order to reduce the occurrence of infectious diseases. Thus, the growing initiatives to create healthcare awareness are expected to generate increased demand for SSI prevention products in healthcare settings. This, in turn, is expected to provide opportunities to product manufacturers to invest and grow in the market.
Surgical Site Infection Market Competitive Landscape
As part of business strategy, key players are undergoing mergers and acquisitions to increase their market share. For instance, in June 2016, B Braun Medical, the South African subsidiary of B Braun Melsungen AG, completed the acquisition of Dismed Pharmaceutical (Pty.) Ltd. and Dismed Criticare (Pty.) Ltd. The acquisition of Dismed’s hand sanitizer and intravenous fluid businesses helped B Braun Medical to cater to its customers in South Africa with its offering of a well-rounded portfolio of effective medical care solutions.
Some of the other key players operating in the surgical site infection market are 3M Company, STERIS plc, Stryker Corporation, Medtronic plc, Prescient Surgical Inc., Covalon Technologies Ltd., Surgical Site Infection Prevention LLC, HARTMANN GROUP, Mölnlycke Health Care AB, and Becton, Dickinson and Company.
Want a report tailored exactly to your business strategy?
Request CustomizationWant an insight-rich discussion with the report author?
Speak to AnalystOur dedication to providing the most-accurate market information has earned us verification by Dun & Bradstreet (D&B). We strive for quality checking of the highest level to enable data-driven decision making for you
Our insights into the minutest levels of the markets, including the latest trends and competitive landscape, give you all the answers you need to take your business to new heights
With 24/7 research support, we ensure that the wheels of your business never stop turning. Don’t let time stand in your way. Get all your queries answered with a simple phone call or email, as and when required
We take a cautious approach to protecting your personal and confidential information. Trust is the strongest bond that connects us and our clients, and trust we build by complying with all international and domestic data protection and privacy laws