Report Code: 11385 | Available Format: PDF | Pages: 176
Hospital-Acquired Infection (HAI) Diagnostics Market by Product (Reagents and Consumables, Instruments), by Test Type (Immunoassay, Molecular Diagnostics, Urinalysis), by Application (Drug-Resistance, Disease), by Infection Type (UTIs, Surgical Site, Bloodstream, Gastrointestinal Hospital Acquired Pneumonia), by End User (Hospitals, Clinics, ASCs), by Geography (U.S., Canada, Mexico, Germany, France, U.K., Italy, Spain, Japan, China, Australia, South Korea, India, Brazil, Saudi Arabia, South Africa), Global Market Size, Share, Development, Growth, and Demand Forecast, 2016-2023
- Report Code: 11385
- Available Format: PDF
- Pages: 176
- Report Description
- Table of Contents
- Market Segmentation
- Request Free Sample
Hospital-Acquired Infection Diagnostics Market Overview
The global hospital-acquired infection (HAI) diagnostics market size is estimated to be valued at $ 2,657.6 million in 2016, and it is forecasted to grow at a CAGR of 7.6% during 2017–2023. In terms of both size and growth, the category of reagents and consumables is estimated to account for a larger share in 2017 and is expected to lead the market in the coming years as well.
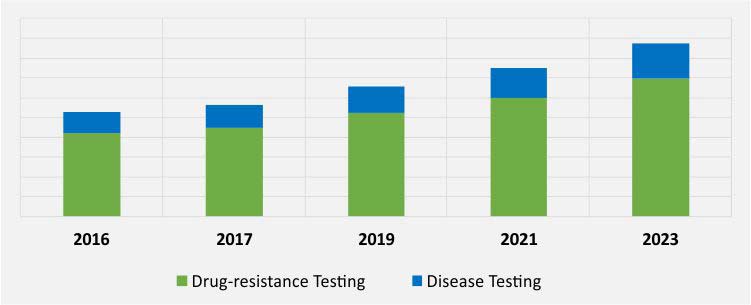
GLOBAL HOSPITAL-ACQUIRED INFECTION DIAGNOSTICS MARKET, BY APPLICATION, $M (2016–2023)
By test type, the molecular diagnostics is expected to witness the fastest growth in demand, registering a CAGR of 12.9% during the forecast period. The factors such increase in the prevalence of the infectious diseases, increase in awareness and acceptance of personalized medicines, and growth in the biomarker identification drive the growth of this market. In addition, increase in adoption of the molecular diagnostics in the emerging economies act as an opportunity for the market growth.
Disease testing is an application of HAI diagnostics, expected to witness the faster growth in demand, with a CAGR of 7.9% during the forecast period. With the technological advancement in medical field, there have been emergence of many modern diagnostic techniques and instruments that has made the diagnosis process more accurate and easy. Technological breakthrough has also led to the development of flow cytometry, mass spectrometry, near-infrared spectroscopy, nucleic acid-based technologies, isothermal microcalorimetry that are widely adopted for infectious disease diagnosis. Hence, increase in prevalence of infectious disease and technological advancement drives the market growth.
Urinary tract infections (UTIs) is estimated to lead the HAI diagnostics market in terms of size, generating $ 829.0 million revenue in 2017, and is also projected to reach a significant growth accounting $1,337.1 million by 2023. Hospital acquired urinary tract infections occur due to medical devices, for instance, various urinary tract infections are caused due to improper use of catheters. According to Centers for Disease Control and Prevention (CDC) 75% of hospital acquired UTIs are associated with a urinary catheter. Additionally, between 15-25% of hospitalized patients receive urinary catheters during their hospital stay.
By end user, the hospitals are estimated to account for the largest share, in terms of value, in the HAI diagnostics market in 2017, across the globe. However, ambulatory surgery centers (ASCs) are estimated to witness the fastest growth in demand, registering a CAGR of 7.9% during the forecast period. The emergence of ASCs is due to the development of point-of-care medical sensors for monitoring and diagnosis outside hospitals. Hence, with the development of ASC the HAI diagnostics market is expected to witness high growth.
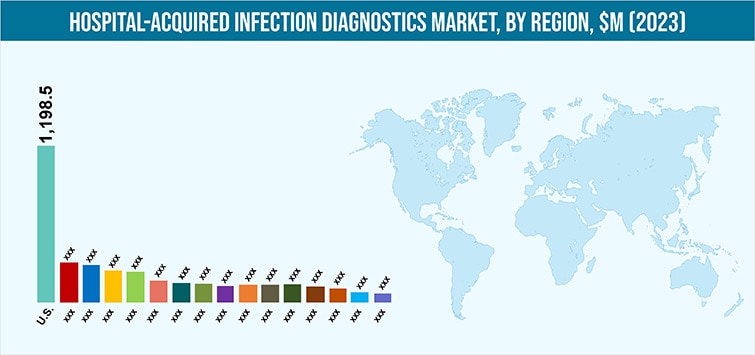
Globally, North America is estimated to account for the largest share, in terms of value, in the HAI diagnostics market in 2017, and is also estimated to lead during the forecast period, accounting $ 1,635.1 million by 2023. The leading position of the region is attributed to its well-developed healthcare industry, presence of leading hospitals acquired infection diagnostics manufacturers in the region, and rise in number of patient’s suffering from nosocomial infections.
Hospital-Acquired Infection Diagnostics Market Dynamics
Growth Drivers
Factors driving the growth of the HAI diagnostics industry are the rising geriatric population base, demand for HAI diagnostics from middle and low-income countries, and development of technologically advanced diagnostic products. The rise in geriatric population will eventually boost the growth of HAI diagnostics market, for instance, according to population reference bureau, the number of Americans ages 65 and older is projected to more than double from 46 million in 2017 to over 98 million by 2060, and the 65-and-older age group’s share of the total population will rise to nearly 24 percent from 15 percent.
Additionally, the development of technologically advanced diagnostic products aids by offering accurate and reliable results with low turnaround time. Major companies are investing in the development of the molecular diagnostics. For instance, in April 2017, F. Hoffmann-La Roche AG, launched cobas Liat PCR system with four assays, which also include 20-minute real-time PCR nucleic acid test to detect Clostridium difficile.
Opportunities
There are many awareness programs introduced by various governmental and nonprofit organizations to reduce the loss of lives globally, that is expected to provide ample opportunities for the growth of the HAI diagnostics market. For instance, according to CDC, about 6–7% of population lose their lives due to hospital acquired infections in developed countries, whereas in developing countries about 10–12% population are affected by hospital acquired infections. World Health Organization (WHO) had introduced an awareness program known as Patient Safety Solutions, in which WHO promoted hand hygiene adherence as a health care facility on priority to reduce the burden of infectious diseases.
-Diagnostics-Market.jpg)
Hospital-Acquired Infection Diagnostics Market Competitive Landscape
As part of the business strategy, major market players are focusing on undergoing collaborative agreements to increase their HAI diagnostics market share in the coming years. For instance, in July 2017, Abbott Laboratories entered into an agreement with Bigfoot Biomedical, Inc. to develop and sell diabetes management system in North America. As part of the agreement, both the companies will develop and commercialize diabetes management systems, integrating Abbott's FreeStyle Libre glucose sensing technology with Bigfoot's insulin delivery solutions in the United States.
Similarly, in March 2017, Sanofi and Sanofi Pasteur, vaccines business unit of Sanofi, collaborated with MedImmune, a subsidiary of AstraZeneca plc, for the development of a monoclonal antibody (mAb), MEDI8897, which prevents the growth of respiratory syncytial virus (RSV).
Want a report tailored exactly to your business strategy?
Request CustomizationWant an insight-rich discussion with the report author?
Speak to AnalystOur dedication to providing the most-accurate market information has earned us verification by Dun & Bradstreet (D&B). We strive for quality checking of the highest level to enable data-driven decision making for you
Our insights into the minutest levels of the markets, including the latest trends and competitive landscape, give you all the answers you need to take your business to new heights
With 24/7 research support, we ensure that the wheels of your business never stop turning. Don’t let time stand in your way. Get all your queries answered with a simple phone call or email, as and when required
We take a cautious approach to protecting your personal and confidential information. Trust is the strongest bond that connects us and our clients, and trust we build by complying with all international and domestic data protection and privacy laws