Report Code: 10539 | Available Format: PDF
Hepatitis Drugs Market Revenue Forecast Report: Size, Share, Recent Trends, Strategic Developments, Segmentation Analysis, and Evolving Opportunities, 2024–2030
- Report Code: 10539
- Available Format: PDF
- Report Description
- Table of Contents
- Request Free Sample
Market Overview
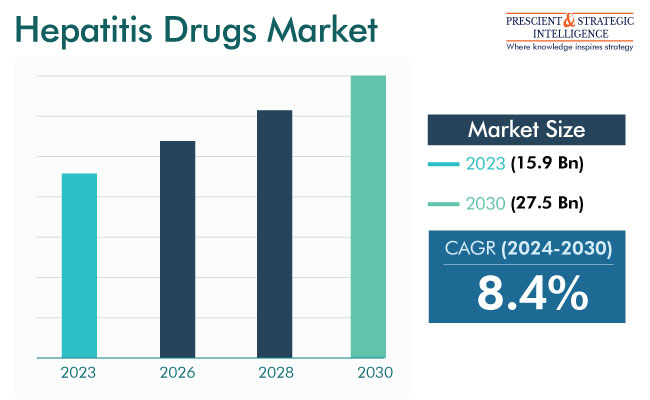
The hepatitis drugs market will propel from USD 15.9 billion (E) in 2023 to USD 27.5 billion in 2030, at 8.4% CAGR between 2024 and 2030.
The growth of this industry is because of the increasing incidence of hepatitis, the surging elderly population, and the rising investments by private and public organizations in research and development for new drugs.
Hepatitis is the inflammation of the liver caused by various infectious and non-infectious agents. Untreated, it can even prove fatal. The consumption of alcohol or even some medicines can also cause hepatitis. In fact, many medicines are toxic to the liver, which is why a doctor’s prescription is advised.
A study conducted by the World Health Organization found that an estimated 4.5 million premature deaths from hepatitis can be prevented in LMICs by 2030 through educational campaigns, diagnostic tests, vaccination, and medicines. Moreover, the WHO aims to decrease new hepatitis deaths by 65% and infection cases by 90% by 2030. Its campaign on World Hepatitis Day helps governments across the globe in raising awareness regarding this condition.
Regulatory Approvals and New Drugs Launches To Boost Market
The increasing number of regulatory approvals for hepatitis drugs is driving the growth of the industry. For instance, in June 2021, the U.S. FDA approved Epclusa (sofosbuvir/velpatasvir) for treating chronic hepatitis C infection in children as young as 3 years of age, regardless of the severity or genotype.
Furthermore, in March 2023, Lupin Limited announced the U.S. FDA approval for its Abbreviated New Drug Application (ANDA) for tenofovir alafenamide tablets. This tablet is under investigation for treating chronic hepatitis B. Additionally, in September 2020, MYR GmbH introduced HEPCLUDEX (bulevirtide) in France, Austria, and Germany to treat chronic hepatitis D and B.
Rising Incidence of Different Kinds of Hepatitis Is Key Driver
The rising count of cases of different types of hepatitis, especially C and B, is playing a key role in driving the industry growth. As per the Hepatitis B Foundation, chronic hepatitis C and B cause 80% of all the liver cancer cases across the globe, and they are the second-most-common cause of cancer deaths.
Moreover, as per the WHO, an estimated 58 million individuals are infected with chronic hepatitis C virus, with around 1.5 million new infections occurring every year. There are about 3.2 million children and adolescents with this infection.
Furthermore, in 2019, about 296 million individuals were living with chronic hepatitis B infection. It also led to about 820,000 mortalities in 2019, mostly from hepatocellular carcinoma and cirrhosis. Therefore, the surging occurrence of these two strains of the infection has increased the requirement for medications.
Additionally, the increasing participation of government associations and players, in the form of R&D funding and promotion of early diagnostic testing, is helping in the industry’s expansion. This is because such initiatives are promoting diagnosis, offering customized solutions to patients, and supporting the advancement of drug therapies.
Mergers & Acquisitions among Pharmaceutical Firms Are Major Trend
Mergers & acquisitions are trending among the players operating in this industry. The major companies are actively executing such strategies to offer new solutions for hepatitis treatment. Moreover, such strategic developments are focused on accelerating and streamlining the process of drug development, to offer better options for treating this condition.
For instance, in March 2021, Gilead Sciences Inc., a biopharmaceutical company based in the U.S., announced the acquisition of MYR GmbH for a cash consideration of USD 1.55 billion. This acquisition offers Gilead Sciences Inc. Hepcludex (bulevirtide), a medication for treating chronic hepatitis D virus in adults with compensated liver disease.
| Report Attribute | Details |
Market Size in 2023 |
USD 15.9 billion (E) |
Revenue Forecast in 2030 |
USD 27.5 billion |
Growth Rate |
8.4% CAGR |
Historical Years |
2017-2023 |
Forecast Years |
2024-2030 |
Report Scope |
Market Trends, Drivers, and Restraints; Revenue Estimation and Forecast; Segmentation Analysis; Impact of COVID-19; Companies’ Strategic Developments; Market Share Analysis of Key Players; Company Profiling |
Explore more about this report - Request free sample
Hepatitis C Is Leading Type
The hepatitis C category, based on type, is the largest contributor to the hepatitis drugs industry. This is primarily because of the increase in the incidence of this infection across developed and developing regions. Apart from this, the rising availability of effective drugs is likely to boost the expansion of this category during the forecast period.
In addition, the rising number of clinical studies regarding hepatitis C diagnosis and treatment is helping the category grow. For example, Task Force for Global Health sponsored a clinical trial in February 2022 to evaluate the results of mother–infant pairs exposed to direct-acting antivirals during pregnancy. Such medicines can cure over 95% of the persons with hepatitis C infection, but access to treatment and diagnosis is low.
Continued Dominance of Drugstores and Retail Pharmacies
Throughout this decade, the drugstores/retail pharmacies category is likely to dominate the end user segment. Because almost all the medications used to treat this condition are available on a prescription, these stores are preferred by patients. Pharmacies also employ learned professionals, which counsel patients on the correct dosage and usage directions.
North America Is Market Leader
North America is leading the industry, mainly because of the rising accessibility of drugs and increasing prevalence of this infections in the region. Moreover, the advantageous reimbursement policies and accessibility of developed healthcare infrastructure are supporting the expansion of the industry.
Furthermore, the U.S. is implementing certain strategic plans for enhancing the viral-hepatitis-related health outcomes of patients, improving disease data usage and surveillance, and reducing disparities. In addition, the existence of major companies, the swift introduction of new products, and the high number of awareness programs will support the progress of the North American industry.
For example, in March 2022, VBI Vaccines Inc. declared the availability of PreHevbrio, a recombinant vaccine, in the U.S. This vaccine is used to prevent infection caused by all known hepatitis B virus subtypes in people 18 years or older. The U.S. FDA had approved PreHevbrio in November 2021.
APAC will witness the fastest growth during the projection period. This can be primarily because of the high disease burden, developments in sanitation and healthcare, improvements in the knowledge of hepatitis vaccination, and increase in the access to medications.
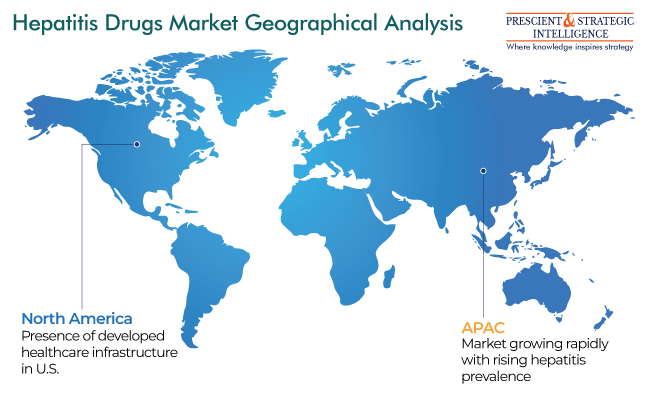
Moreover, countries such as China, India, and Indonesia are constantly increasing their investment to allow easier access to high-quality healthcare. In addition, the increase in the requirement for generic medicines will support the growth of the hepatitis drugs industry in APAC till the end of this decade.
Key Companies in Hepatitis Drugs Market
- Dynavax Technologies Corporation
- Gilead Sciences Inc.
- F. Hoffmann-La Roche Ltd.
- Biocon Ltd.
- AbbVie Inc.
- Johnson & Johnson Services Inc.
- Merck & Co. Inc.
- Cipla Inc.
- Novartis AG
- Mitsubishi Chemical Corporation
- AbbVie Inc.
- GlaxoSmithKline plc
- Lupin Ltd.
- Merck & Co. Inc.
- Teva Pharmaceutical Industries Ltd.
- Bristol–Myers Squibb Company
Want a report tailored exactly to your business strategy?
Request CustomizationWant an insight-rich discussion with the report author?
Speak to AnalystOur dedication to providing the most-accurate market information has earned us verification by Dun & Bradstreet (D&B). We strive for quality checking of the highest level to enable data-driven decision making for you
Our insights into the minutest levels of the markets, including the latest trends and competitive landscape, give you all the answers you need to take your business to new heights
With 24/7 research support, we ensure that the wheels of your business never stop turning. Don’t let time stand in your way. Get all your queries answered with a simple phone call or email, as and when required
We take a cautious approach to protecting your personal and confidential information. Trust is the strongest bond that connects us and our clients, and trust we build by complying with all international and domestic data protection and privacy laws