Report Code: 11362 | Available Format: PDF | Pages: 124
Artificial Disc Market Research Report: By Type (Cervical Disc, Lumbar Disc), Material (Metal-on-Polymer) - Global Industry and Forecast to 2024
- Report Code: 11362
- Available Format: PDF
- Pages: 124
- Report Description
- Table of Contents
- Market Segmentation
- Request Free Sample
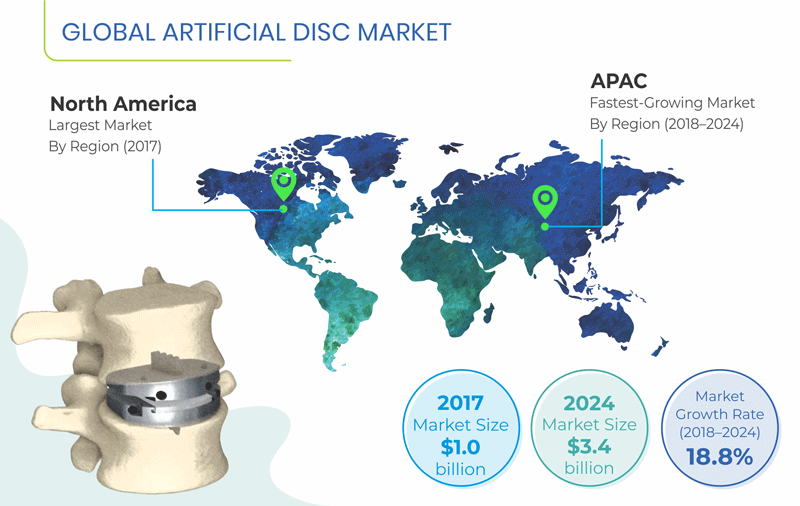
Valued at $1.0 billion in 2017, the global artificial disc market is projected to reach $3.4 billion by 2024, witnessing a CAGR of 18.8% between 2018–2024.
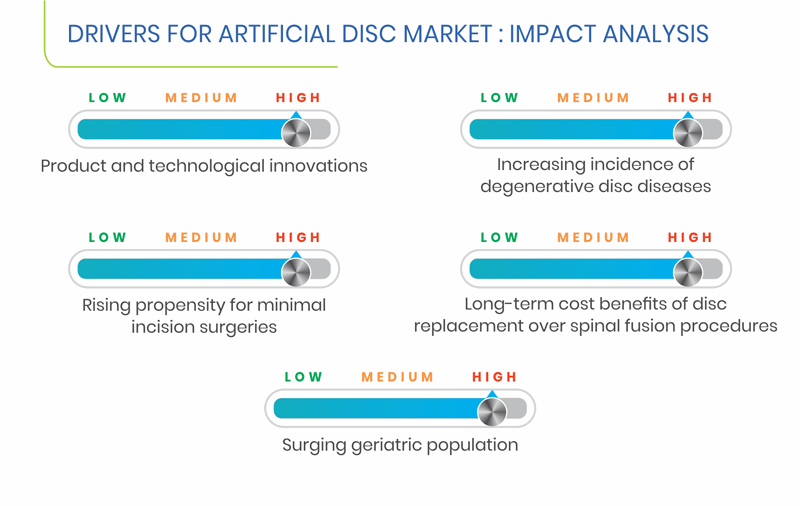
Among all regions, Asia-Pacific (APAC) is expected to witness the fastest growth in the market, during the forecast period. This can be mainly ascribed to the growing geriatric population, rising healthcare expenditure, and increasing investments by public and private organizations in the healthcare sector to cater to unmet medical needs of the people in the region. According to the United Nations Department of Economic and Social Affairs (UNDESA) report entitled World Population Ageing 2015, in Japan, 23.3% of the population was aged 60 years and above in 2000, and it is estimated to reach 37.3% by 2030. As the elderly population is more susceptible to develop lumbar- and cervical-related diseases, the artificial disc market is projected to increase in the region.
Dynamics of Artificial Disc Market
The key trend observed in the artificial disc market is the increasing number of mergers and acquisitions among the key players to expand their geographical footprint and consumer base. For instance, in July 2016, Zimmer Biomet Holdings Inc. acquired LDR Holding Corporation (LDR), a medical device company dedicated to designing and commercializing surgical technologies for the treatment of patients suffering from spine disorders. The acquisition aimed at strengthening the position of Zimmer Biomet Holdings Inc. in musculoskeletal healthcare with the addition of a premier spine platform to its product portfolio.
Long-term cost benefits of disc replacement over spinal fusion procedures is a key factor driving the growth of the artificial disc market, globally. Spinal fusion requires various surgeries over a span of time, whereas artificial disc replacement is a one-time surgical procedure. According to a study published in the North American Spine Society (NASS) meeting in 2013 by researchers of the Rothman Institute and the Texas Back Institute, the total costs for average of 48 months were lower in the patients receiving cervical arthroplasty (artificial disc replacement), around $34,000, as against $39,000 in the patients receiving anterior cervical discectomy and fusion (ACDF).
An increasing number of clinical studies on artificial discs over ACDF surgeries have been observed, globally. For instance, in November 2016, Spineart SA in its clinical data for Baguera C cervical disc prosthesis concluded that cervical disc replacement with Baguera C is a safe and effective procedure for the treatment of radiculopathy due to soft disc herniation or moderate uncarthrosis on one or two levels. Since the studies have shown promising results with the use of these discs, the opportunities for the key players have significantly increased in the artificial disc market to receive regulatory approvals for the commercialization of these products.
Structural Analysis of Artificial Disc Market
The artificial disc market when segmented on the basis of type was dominated by the cervical category, in 2017. The market growth in this category is attributed to the rising prevalence of cervical-related health issues, globally.
The metal-on-polymer material category is expected to be the faster growing category during the forecast period. The popularity of this material can be ascribed to its shock absorption feature.
| Report Attribute | Details |
Historical Years |
2014-2017 |
Forecast Years |
2018-2023 |
Market Size by Segments |
Type, Material |
Market Size of Geographies |
U.S., Canada, Germany, France, U.K., Italy, Spain, Japan, China, India, Brazil |
Explore more about this report - Request free sample
Global Scenario of Artificial Disc Market
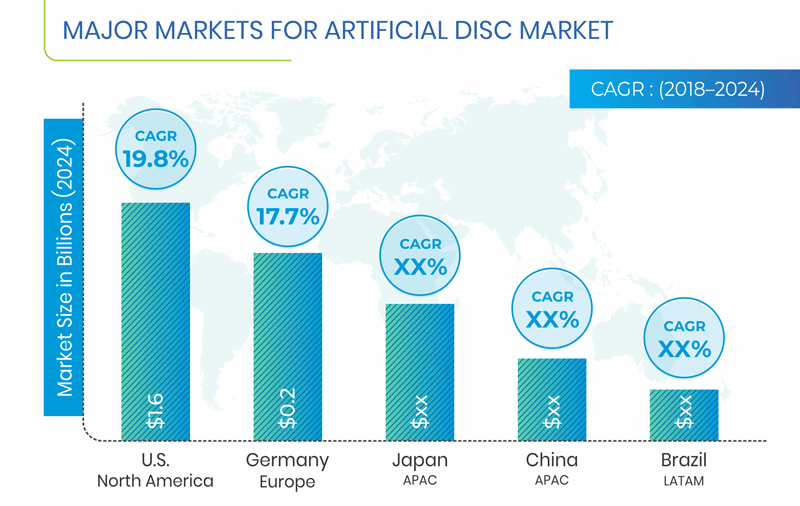
North America is expected to hold the largest share in the global artificial disc market in 2024. This can be mainly attributed to surging geriatric population, rising healthcare expenditure, improving economy, expanding private healthcare sector, and growing awareness among the patients about the availability of various treatment options. Further, considering the market dynamics, the U.S. is expected to dominate the North American market.
The APAC region is expected to be fastest growing region for artificial disc market, mainly on account of rising healthcare expenditure, increasing per capita income, coupled with surge in geriatric population. Moreover, Japan is projected to account for the largest market share in the APAC region.
Competitive Landscape of Artificial Disc Market
The artificial disc market is fragmented in nature with the presence Medtronic plc, Johnson & Johnson, Zimmer Biomet Holdings Inc., Globus Medical Inc., B. Braun Melsungen AG, Simplify Medical Inc., NuVasive Inc., Spinal Kinetics Inc., K2M Group Holdings Inc., and AxioMed LLC.
Recent Strategic Developments of Artificial Disc Market
In the recent years, major players in the artificial disc market have taken strategic measure of product approvals and launches, partnerships, and acquisitions to gain a significant share. For instance, in January 2018, NuVasive Inc. acquired SafePassage, an intraoperative neurophysiological monitoring (IONM) service provider, with the aim to strengthen company’s IONM business line.
Moreover, in March 2017, NuVasive Inc. received the USFDA 510(k) clearance for its CoRoent Small Interbody System, indicated for intervertebral body fusion at multiple contiguous levels in the cervical spine. The product approval marked the first U.S. clearance for a cervical cage to be used at up to four contiguous levels.
Artificial Disc Market Size Breakdown by Segment
The Artificial disc market report offer comprehensive market segmentation analysis along with market estimation for the period 2014–2024.
Based on Type
- Cervical Disc
- Lumbar Disc
Based on Material
- Metal-on-Polymer
- Others (Metal-on-Metal, Metal-on-Ceramic, and Polymer-on-Ceramic)
Geographical Analysis
- North America
- U.S.
- Canada
- Europe
- Germany,
- France
- U.K.
- Italy
- Spain
- Asia-Pacific (APAC)
- Japan
- China
- India
- Latin America (LATAM)
- Brazil
- Middle East & Africa (MEA)
Want a report tailored exactly to your business strategy?
Request CustomizationWant an insight-rich discussion with the report author?
Speak to AnalystOur dedication to providing the most-accurate market information has earned us verification by Dun & Bradstreet (D&B). We strive for quality checking of the highest level to enable data-driven decision making for you
Our insights into the minutest levels of the markets, including the latest trends and competitive landscape, give you all the answers you need to take your business to new heights
With 24/7 research support, we ensure that the wheels of your business never stop turning. Don’t let time stand in your way. Get all your queries answered with a simple phone call or email, as and when required
We take a cautious approach to protecting your personal and confidential information. Trust is the strongest bond that connects us and our clients, and trust we build by complying with all international and domestic data protection and privacy laws